石灰化大動脈弁狭窄症
Calcific aortic stenosis
2016年3月3日 Nature Reviews Disease Primers Article number: 16006 (2016) doi: 10.1038/nrdp.2016.6
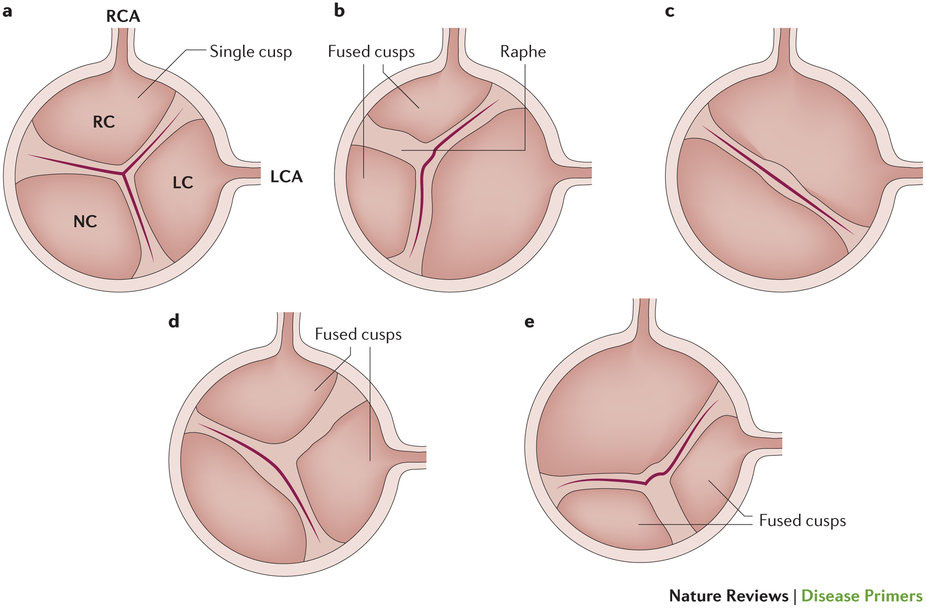
石灰化大動脈弁狭窄症(AS)は先進国で最もよくみられる心臓弁障害である。本症は進行性の線維石灰化リモデリングおよび大動脈弁尖肥厚を特徴とし、数年かけて進展すると心臓流出路の重度閉塞が引き起こされる。先進国においてASは、冠動脈疾患と全身性動脈性高血圧に次いで3番目に頻度の高い心血管疾患であり、その罹患率は一般集団で0.4%、65歳以上では1.7%である。先天性異常(二尖弁)および高齢は、石灰化ASの強力なリスク因子である。メタボリックシンドロームおよび血漿リポタンパク質濃度の上昇も石灰化ASのリスク上昇と関連している。石灰化ASの病理生物学は複雑であり、遺伝的要因、リポタンパク質の沈着と酸化、慢性炎症、心臓弁間質細胞から骨芽細胞への変換および顕著な弁尖石灰化が関係している。ASの進行抑制に有効であることが示されている薬物療法はないが、有望な治療標的として、リポタンパク質(a)、レニン-アンジオテンシン系、NF-κBリガンド受容体活性化因子(RANKL、別名:TNFSF11)およびエクトヌクレオトダーゼがある。現在、重症ASの唯一の有効な治療法は大動脈弁置換術(AVR)である。ASの診断と病期分類は、ドプラ心エコー法による狭窄の重症度および左室機能の評価ならびに症状の有無に基づいて行われる。この10年間で経カテーテルAVRが導入されたことは、外科的弁置換を行うには高リスクまたは非常に大きなリスクを有する患者にとって、斬新な治療革新であり、近い将来、この新しい技術は低リスク患者にも拡大されるだろう。
PrimeView
石灰化大動脈弁狭窄症(AS)は、弁尖の肥厚と石灰化、およびそれに続発する大動脈弁を介する血流障害によって発症する。このPrimeViewでは、唯一の治療法である大動脈弁置換術によるASの管理法について取りまとめる。
本Primerの図解サマリー

