Research Abstract
膜内KcsAカリウムチャネルの開ゲート構造を細胞質側から見る
The Open Gate Structure of the Membrane-Embedded KcsA Potassium Channel Viewed From the Cytoplasmic Side
2013年1月15日 Scientific Reports 3 : 1063 doi: 10.1038/srep01063
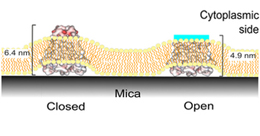
チャネルタンパクの結晶解析により、イオンチャネル開閉の分子機構に手がかりが得られているが、これらの構造は膜から取り出された状態のものであり、未解明の部分構造も残されている。今回、膜に埋め込まれた状態でのKcsAカリウムチャネルの活性化ゲートの構造を、原子間力顕微鏡(AFM)を使って明らかにした。KcsAチャネルの活性化ゲートは細胞内側にあり、この部分を明瞭に観察するため細胞内ドメインを切除した。チャネル四量体が開構造をとった際の個々のサブユニットおよび中央に開いた孔を、像としてとらえることができた。さらに、これまでとらえられなかったチャネル入口に突き出したへリックスもとらえられた。分子動力学シミュレーションによれば、この突出へリックスは、ゲート開口部で大きく揺らいでいる。この揺らぎが、単一チャネル記録では激しい開チャネルノイズとして観測された。ゲート構造におけるこの突出へリックスの役割について論じる。
角野 歩1, 2, 炭竃 享司3, 岩本 真幸3, 出羽 毅久1, 4 & 老木 成稔3
- 名古屋工業大学大学院 工学研究科 未来材料創成工学専攻
- 日本学術振興会 特別研究員
- 福井大学 医学部医学科 分子生理学研究室
- 独立行政法人 科学技術振興機構 戦略的創造研究推進事業 さきがけ(JST-PRESTO)
Crystallographic studies of channel proteins have provided insight into the molecular mechanisms of ion channels, even though these structures are obtained in the absence of the membrane and some structural portions have remained unsolved. Here we report the gating structure of the membrane-embedded KcsA potassium channel using atomic force microscopy (AFM). The activation gate of the KcsA channel is located on the intracellular side, and the cytoplasmic domain was truncated to clear the view of this location. Once opened, the individual subunits in the tetramer were resolved with the pore open at the center. Furthermore, AFM was able to capture the previously unsolved bulge helix at the entrance. A molecular dynamics simulation revealed that the bulge helices fluctuated dramatically at the open entryway. This dynamic behavior was observed as vigorous open-channel noise in the single-channel current recordings. The role of the bulge helices in the open gate structure is discussed.

