C-N結合の切断を経由するStilleカップリング
Stille coupling via C–N bond cleavage
2016年9月30日 Nature Communications 7 : 12937 doi: 10.1038/ncomms12937
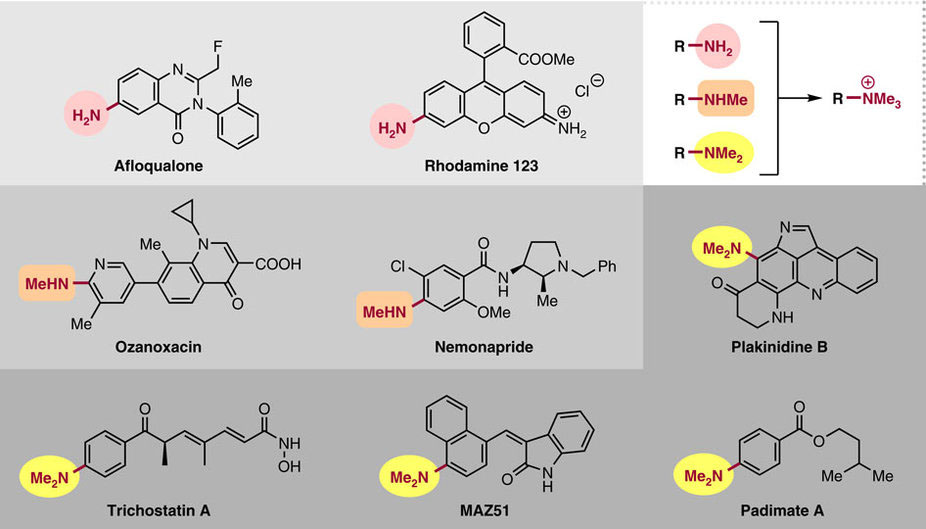
クロスカップリングは、機能性分子の合成における基本的な反応であり、例えば、フェノール、アニリン、アルコール、アミンや、それらの誘導体などに広く適用されている。今回我々は、C-N結合の切断を経由する、第四級アンモニウム塩のNi触媒Stilleクロスカップリング反応について報告する。触媒量の市販のNi(cod)2、イミダゾール配位子、3.0当量のCsFの存在下で、アリール/アルキルトリメチルアンモニウム塩[Ar/R-NMe3]+がアリールスタンナンとモル比1:1で穏やかに反応し、広い官能基許容性を持つ対応するカップリング生成物が得られる。検証実験や理論計算とともに、Niを含む中間体の単離とX線結晶解析を行い、C-N結合切断段階を含む反応経路を提案する。本反応は、アミン/アニリンを足掛かりとして、様々なπ共役系化合物への変換に広く適用できるツールとなることが期待される。
Corresponding Authors
Cross-coupling is a fundamental reaction in the synthesis of functional molecules, and has been widely applied, for example, to phenols, anilines, alcohols, amines and their derivatives. Here we report the Ni-catalysed Stille cross-coupling reaction of quaternary ammonium salts via C–N bond cleavage. Aryl/alkyl-trimethylammonium salts [Ar/R–NMe3]+ react smoothly with arylstannanes in 1:1 molar ratio in the presence of a catalytic amount of commercially available Ni(cod)2 and imidazole ligand together with 3.0 equivalents of CsF, affording the corresponding biaryl with broad functional group compatibility. The reaction pathway, including C–N bond cleavage step, is proposed based on the experimental and computational findings, as well as isolation and single-crystal X-ray diffraction analysis of Ni-containing intermediates. This reaction should be widely applicable for transformation of amines/quaternary ammonium salts into multi-aromatics.

