Research Abstract
中心に窒素原子をもつバッキーボウルおよびそのC60会合体
Nitrogen-embedded buckybowl and its assembly with C60
2015年9月4日 Nature Communications 6 : 8215 doi: 10.1038/ncomms9215
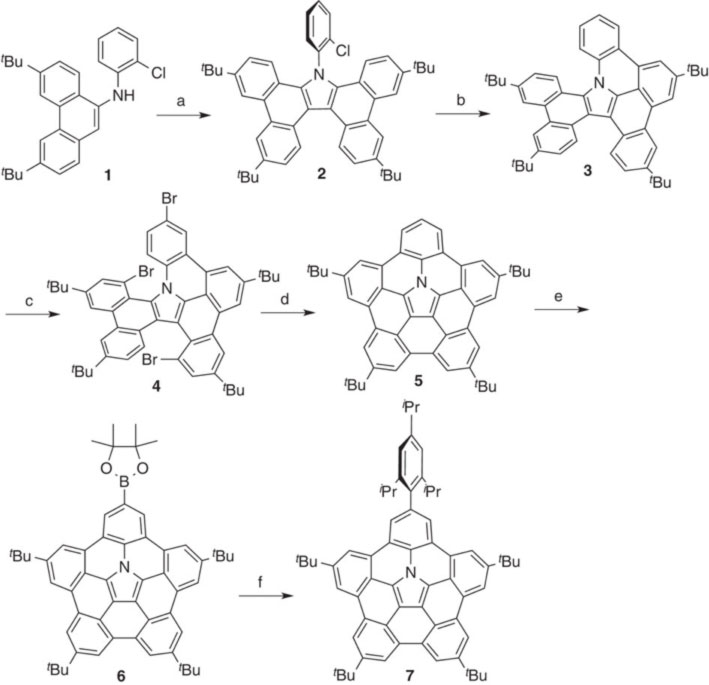
曲面π共役分子は、湾曲したπ面に起因するユニークな特性をもつため、大きな関心を集めている。しかし、そうした歪んだ分子の合成には過酷な反応条件が必要となるため、ヘテロ原子を含む曲面π系の合成は容易ではない。今回我々は、中央部に窒素をもつπ拡張アザコラニュレン(アザバッキーボウル)の合成について報告する。我々は、9-アミノフェナントレンの酸化によって得たテトラベンゾカルバゾールを、パラジウム触媒による分子内カップリング反応でアザバッキーボウルに変換することに成功した。アザバッキーボウルは、高い電子供与性とπ曲面構造をもつため、溶液中および固体状態でC60と強く会合する。また、得られたアザバッキーボウル/C60集合体において、高い電荷移動度が観測された。アザバッキーボウルは、フラーレンのホスト分子、異方性πドナー分子、窒素含有ナノカーボン材料の前駆体としての応用が期待される。
Hiroki Yokoi, Yuya Hiraoka, Satoru Hiroto, Daisuke Sakamaki, Shu Seki & Hiroshi Shinokubo
Corresponding Authors
Curved π-conjugated molecules have attracted considerable interest because of the unique properties originating from their curved π surface. However, the synthesis of such distorted molecules requires harsh conditions, which hamper easy access to heteroatom-containing curved π systems. Here we report the synthesis of a π-extended azacorannulene with nitrogen in its centre. The oxidation of 9-aminophenanthrene provides tetrabenzocarbazole, which is converted to the azabuckybowl through palladium-catalysed intramolecular coupling. The electron-donating nature and curved π surface of the azabuckybowl enable its tight association with C60 in solution and solid states. High charge mobility is observed for the azabuckybowl/C60 assembly. This compound may be of interest in the fields of curved π systems as fullerene hosts, anisotropic π donors and precursors to nitrogen-containing nanocarbon materials.

