Research Abstract
マウスの刷り込み型X染色体の不活性化と胚発生に、母系染色体特異的なH3K9me3修飾が果たす役割
The role of maternal-specific H3K9me3 modification in establishing imprinted X-chromosome inactivation and embryogenesis in mice
2014年11月14日 Nature Communications 5 : 5464 doi: 10.1038/ncomms6464
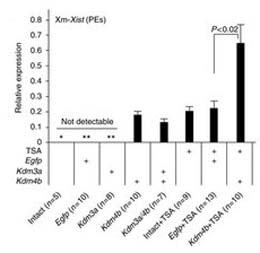
マウスの胚の発生には、Xist遺伝子を抑制して1本のX染色体の活性を維持することが重要である。Xistの活性化因子RNF12/RLIMは卵子由来の因子として存在するが、母系Xist(Xm-Xist)は着床前に抑制され、父系Xistのみが活性化されて刷り込み型X染色体不活性化(XCI)が確立する。今回我々は、着床前の胚のクロマチン解析が簡単に行える再現性の高いクロマチン免疫沈降法を用いて、Xistのプロモーター領域にはH3K9me3修飾が多く見られ、RNF12によるXm-Xistの活性化を妨げていることを明らかにした。胚性幹(ES)細胞ではXistのプロモーター領域のH3K9me3修飾のレベルが低下しており、ES由来のクローン胚では、RNF12に依存してXistが発現する。さらに、単為発生胚では70~80%が着床後すぐに致死となるが、その主な原因は、卵子で刷り込みを受けた遺伝子が父系染色体から発現されないためではなく、栄養外胚葉でXm-XCIが起こらないためであった。今回の研究によって、H3K9me3が、Xm-Xist抑制のインプリンティングに関わっていることが分かり、胚の発生に母系特異的なH3K9me3が大きな役割を果たしていることが明らかになった。
福田 篤1, 冨川 順子2, 三浦 巧1, 秦 健一郎2, 中林 一彦2, ケビン イーガン3, 阿久津 英憲1 & 梅澤 明弘1
- 国立成育医療研究センター 再生医療センター 生殖・細胞医療研究部
- 国立成育医療研究センター周産期病態研究部
- ハーバード大学幹細胞研究所(アメリカ)
Maintaining a single active X-chromosome by repressing Xist is crucial for embryonic development in mice. Although the Xist activator RNF12/RLIM is present as a maternal factor, maternal Xist (Xm-Xist) is repressed during preimplantation phases to establish imprinted X-chromosome inactivation (XCI). Here we show, using a highly reproducible chromatin immunoprecipitation method that facilitates chromatin analysis of preimplantation embryos, that H3K9me3 is enriched at the Xist promoter region, preventing Xm-Xist activation by RNF12. The high levels of H3K9me3 at the Xist promoter region are lost in embryonic stem (ES) cells, and ES-cloned embryos show RNF12-dependent Xist expression. Moreover, lack of Xm-XCI in the trophectoderm, rather than loss of paternally expressed imprinted genes, is the primary cause of embryonic lethality in 70–80% of parthenogenotes immediately after implantation. This study reveals that H3K9me3 is involved in the imprinting that silences Xm-Xist. Our findings highlight the role of maternal-specific H3K9me3 modification in embryo development.

