Research Abstract
複数の薬剤を伴った多剤耐性調節因子RamRの結晶構造
RamRは重要な多剤耐性因子だが、その構造とそれが反応する分子はこれまで知られていなかった。本稿で著者らは、複数の薬剤と複合体を形成したRamRの結晶構造を報告し、基質認識部位の非常に高い柔軟性を明らかにした。
The crystal structure of multidrug-resistance regulator RamR with multiple drugs
2013年6月26日 Nature Communications 4 : 2078 doi: 10.1038/ncomms3078
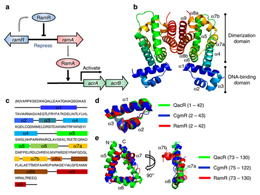
RamRは、多剤排出システム遺伝子acrAB-tolCの発現を制御するRamAタンパク質をコードする遺伝子の転写抑制因子である。RamRは重要な多剤耐性因子であるが、その構造およびそれが反応する分子の正体は知られていなかった。今回我々は、ベルベリン、クリスタルバイオレット、デカリニウム、臭化エチジウムおよびローダミン6Gを含む複数の薬剤と複合体を形成したRamRの結晶構造を報告する。全ての化合物はRamRのPhe155と相互作用しており、またそれぞれの化合物は異なるアミノ酸残基によって取り囲まれていることが分かった。これらの化合物のRamRへの結合はDNA結合親和性を低下させ、その結果ramAの発現が上昇する。我々の結果は、多剤耐性に関与する細菌の排出を調節するRamRの基質認識部位の非常に高い柔軟性を明らかにしたものである。
山崎 優1, 2, 二階堂 英司1, 2, 中島 良介3, 櫻井 啓介3, 藤原 大佑4, 藤井 郁雄4 & 西野 邦彦1
- 大阪大学 産業科学研究所 特別プロジェクト研究部門 感染制御学研究分野
- 大阪大学大学院 薬学研究科
- 大阪大学 産業科学研究所 特別プロジェクト研究部門 生体防御学研究分野
- 大阪府立大学大学院 理学系研究科 生物科学専攻
RamR is a transcriptional repressor of the gene-encoding RamA protein, which controls the expression of the multidrug efflux system genes acrAB-tolC. RamR is an important multidrug-resistance factor, however, its structure and the identity of the molecules to which it responds have been unknown. Here, we report the crystal structure of RamR in complex with multiple drugs, including berberine, crystal violet, dequalinium, ethidium bromide and rhodamine 6G. All compounds are found to interact with Phe155 of RamR, and each compound is surrounded by different amino acid residues. Binding of these compounds to RamR reduces its DNA-binding affinity, which results in the increased expression of ramA. Our results reveal significant flexibility in the substrate-recognition region of RamR, which regulates the bacterial efflux participating in multidrug resistance.

