Research Abstract
原発腫瘍は自然免疫系の情報伝達を変化させて、転移前に転移予定地に血管透過性亢進部位を作る
Primary tumours modulate innate immune signalling to create pre-metastatic vascular hyperpermeability foci
2013年5月14日 Nature Communications 4 : 1853 doi: 10.1038/ncomms2856
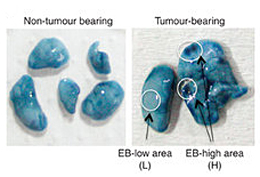
肺転移のマウスモデルでは、明らかな転移巣が出現する前に局所的に血管の透過性の変化が見られ、それが腫瘍の成長の土台となる。しかし、ヒトにもこれが当てはまるかは不明であった。今回我々は、自然免疫応答に重要な役割を担うToll様受容体4の補助受容体MD-2が、C-Cケモカイン受容体CCR2の発現を上昇を伴って、透過性亢進部位の形成のきっかけを作ることをマウスで明らかにした。CCR2-CCL2系は、血清アミロイドA3やS100A8といった透過性亢進誘導因子を大量に分泌させる。MD-2やCCR2を破壊すると透過性亢進部位が形成されなくなり、転移腫瘍細胞の定着が抑えられる。また、遠隔腫瘍のある患者の肺のCCR2発現が上昇している部位には、フィブリノーゲン(透過性亢進が関与する血液凝固の際に出現処理される)も同局在する。これらの知見から考えて、CCR2の発現上昇は、肺がんの転移が起こりやすくなっている領域を示すマーカーになる可能性がある。
平塚 佐千枝1,2, 石橋 幸江1, 富田 毅1, 渡辺 亮3*, 赤司(高村) 祥子4, 村上 正人5**, 鬼島 宏6***,三宅 健介4, 油谷 浩幸3 & 丸 義朗1
- 東京女子医科大学 薬理学教室
- 独立行政法人 科学技術振興機構 さきがけ
- 東京大学 先端科学技術研究センター ゲノムサイエンス分野
- 東京大学医科学研究所 感染・免疫部門 感染遺伝学分野
- 東京大学医科学研究所
- 東海大学 医学部 病理学
*現所属先: 京都大学 IPS細胞研究所
**現所属先: ノバルティス生物医学研究所(スイス)
***現所属先: 弘前大学大学院 医学研究科
In mouse models of lung metastasis, before the appearance of significant metastases, localized changes in vascular permeability have been observed, which appear to set the stage for tumour growth. However, it is unclear whether this is also true in human patients. Here, we show that MD-2, a coreceptor for Toll-like receptor 4 that has a key role in the innate immune response, triggers the formation of regions of hyperpermeability in mice by upregulating C-C chemokine receptor type 2 (CCR2) expression. The CCR2–CCL2 system induces the abundant secretion of permeability factors such as serum amyloid A3 and S100A8. Disruption of MD-2 or CCR2 abrogates the formation of hyperpermeable regions, resulting in reduced tumour cell homing. Furthermore, fibrinogen, which is processed during permeability-mediated coagulation, is also localized in areas of elevated CCR2 expression in tumour-bearing human lungs. Our findings raise the possibility that CCR2 upregulation might represent a marker for regions of increased susceptibility to metastatic homing in lung cancer.

