Research Abstract
細胞外小胞の新しい高純度アフィニティ精製法
A novel affinity-based method for the isolation of highly purified extracellular vesicles
2016年9月23日 Scientific Reports 6 : 33935 doi: 10.1038/srep33935
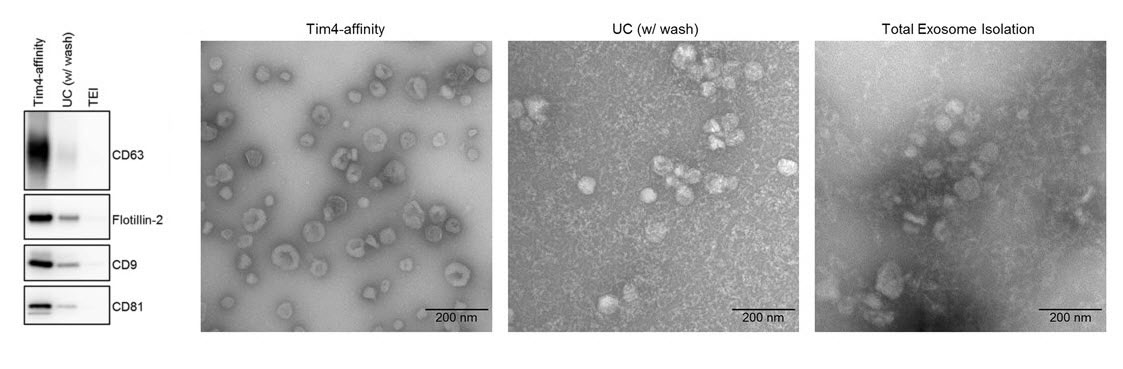
エクソソームやマイクロベジクルなどの細胞外小胞(EV)は、細胞間ネットワークの仲介役として働き、細胞間での細胞成分の交換を可能にしている。EVには、そのEVを産生した細胞に由来する脂質、タンパク質、RNAが含まれるため、細胞種や細胞状態に特異的なバイオマーカーとなる可能性がある。しかし、従来のEV精製法(超遠心法やポリマー沈殿法)には、純度や操作の簡便性の点で問題があった。今回我々は、Tim4タンパク質を利用した新たなEV精製法を開発した。Tim4は、EVの表面に存在するホスファチジルセリンと特異的に結合するが、この結合がCa2+に依存しているため、Ca2+キレート剤を加えることによって、EVをそのままの形でTim4から簡単に遊離させることができる。培養細胞上清や生体液で、このTim4法を用いてEVの精製を行ったところ、従来の方法よりも高純度のEVを得ることができた。Tim4法で精製したEV試料は不純物の含有が少ないため、より多くのEV特異的タンパク質が質量分析により検出でき、さまざまなEVのプロテオームのより良い解析と定量が可能になる。またTim4タンパク質を用いることにより、ELISA法やフローサイトメトリーにおいてEVの効果的な定量が可能となる。このように、EVに対するTim4の親和性は、今後EV研究にさまざまな形で活用されるであろう。
Corresponding Author
Extracellular vesicles (EVs) such as exosomes and microvesicles serve as messengers of intercellular network, allowing exchange of cellular components between cells. EVs carry lipids, proteins, and RNAs derived from their producing cells, and have potential as biomarkers specific to cell types and even cellular states. However, conventional methods (such as ultracentrifugation or polymeric precipitation) for isolating EVs have disadvantages regarding purity and feasibility. Here, we have developed a novel method for EV purification by using Tim4 protein, which specifically binds the phosphatidylserine displayed on the surface of EVs. Because the binding is Ca2+-dependent, intact EVs can be easily released from Tim4 by adding Ca2+ chelators. Tim4 purification, which we have applied to cell conditioned media and biofluids, is capable of yielding EVs of a higher purity than those obtained using conventional methods. The lower contamination found in Tim4-purified EV preparations allows more EV-specific proteins to be detected by mass spectrometry, enabling better characterization and quantification of different EV populations’ proteomes. Tim4 protein can also be used as a powerful tool for quantification of EVs in both ELISA and flow cytometry formats. Thus, the affinity of Tim4 for EVs will find abundant applications in EV studies.

