Research Abstract
かさ高い芳香族アジドの高いクリック反応性の発見
Enhanced clickability of doubly sterically-hindered aryl azides
2011年9月5日 Scientific Reports 1 : 82 doi: 10.1038/srep00082
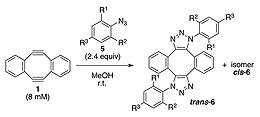
立体的な性質は、分子の反応性を決定する最も基本的な要素の1つである。通常、二分子反応では、反応中心付近のかさ高い置換基が、反応相手の接近を妨げ、結合を形成しにくくする。本論文では、それとは逆に、両オルト位にかさ高い置換基を有する芳香族アジドがアルキンとの無触媒1,3-双極子環化付加反応において著しく高い反応性を示す、という一見すると異常な現象の発見について報告する。意外なことに、2,6-二置換芳香族アジドのアルキンとの反応が、アジド基近傍の大きな立体障害にもかかわらず、無置換フェニルアジドよりも著しく速く進行し、さらには障害のないアルキルアジドよりも速く反応することが明らかになった。実験と理論計算を用いた解析の結果、立体障害によってアジド基と芳香環との共鳴が阻害されることが、この一見矛盾した現象の主な要因であることが示された。これは新しい形式の立体効果による反応の加速法であり、官能基を込み合った環境に戦略的に配置することで、その反応性を高める分子設計が可能であることを示唆している。
- 東京医科歯科大学 大学院疾患生命科学研究部/生体材料工学研究所
- 東京工業大学 大学院生命理工学研究科
- チッソ石油化学株式会社 五井研究所
- 東京工業大学 大学院理工学研究科
Steric character is one of the most fundamental factors to determine the reactivity of the substrate in organic synthesis. In bimolecular reaction, the sterically-bulky group situated close to the reactive center generally prevents the approach of the reaction partner retarding the bond formation. This report describes, to the contrary, significantly enhanced reactivity of 2,6-disubstituted phenyl azides observed in catalyst-free 1,3-dipolar cycloaddition with alkynes, unexpectedly reacting faster than unsubstituted phenyl azide and even more faster than unhindered alkyl azide, despite the steric hindrance adjacent to the reactive azido group. Experimental and computational studies have indicated that the steric hindrance eliciting the inhibition of resonance between azido group and the aromatic ring is the primary cause of this apparently-paradoxical phenomenon. This is the first type of steric acceleration, indicating a possibility of designing a highly reactive functional group by strategically locating it in the sterically-congested environment.

