Research Abstract
Y14の核と細胞質間の往復移動やスプライシングを受けたmRNAへの結合は、Y14の特異的ドメインを介して起こる
Specific Y14 domains mediate its nucleo-cytoplasmic shuttling and association with spliced mRNA
2011年9月14日 Scientific Reports 1 : 92 doi: 10.1038/srep00092
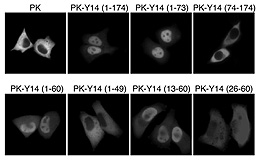
mRNA前駆体のスプライシングによって、mRNA上のエキソンどうしの接合部付近に、エキソン接合部複合体(EJC)と呼ばれるタンパク質複合体が形成される。このEJCの中心部(コア)は、eIF4AIII、MLN51、Y14、Magohという4つのタンパク質で構成される。Y14は核と細胞質の間を行ったり来たりできる核タンパク質で、Magohに特異的に結合し、複合体を形成する。今回我々は、Y14の核局在シグナルが核外への輸送をも司ることを明らかにし、このシグナルをYNSと名付けた。さらに、Y14のカルボキシ末端近くの12アミノ酸からなるペプチドが、スプライシングを受けたmRNAへの結合に必要であり、しかもMagohとの結合にも必要なことを明らかにした。また、Magohと結合ができないY14変異体でも核への局在は起こることから、Magohと結合していないY14が細胞内に存在し、Y14特異的な核内への輸送経路とY14が単独で果たす機能があることが示唆される。
- 東京医科歯科大学 難治疾患研究所
- 京都大学ウイルス研究所
- ペンシルベニア大学医学系大学院(米国)
- 京都大学大学院生命科学研究科
- 京都大学大学院医学研究科
*These authors contributed equally to this work.
Pre-mRNA splicing deposits multi-protein complexes, termed exon junction complexes (EJCs), on mRNAs near exon-exon junctions. The core of EJC consists of four proteins, eIF4AIII, MLN51, Y14 and Magoh. Y14 is a nuclear protein that can shuttle between the nucleus and the cytoplasm, and binds specifically to Magoh. Here we delineate a Y14 nuclear localization signal that also confers its nuclear export, which we name YNS. We further identified a 12-amino-acid peptide near Y14’s carboxyl terminus that is required for its association with spliced mRNAs, as well as for Magoh binding. Furthermore, the Y14 mutants, which are deficient in binding to Magoh, could still be localized to the nucleus, suggesting the existence of both the nuclear import pathway and function for Y14 unaccompanied by Magoh.

