Research Abstract
ディールス・アルダー・スーパージエンを使ってベンゼンをC2H2とC4H4に分解する
A Diels–Alder super diene breaking benzene into C2H2 and C4H4 units
2014年1月8日 Nature Communications 5 : 3018 doi: 10.1038/ncomms4018
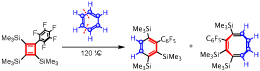
炭素原子6個からなる環状ポリエン(ベンゼン)は非常に安定であるのに対し、炭素原子4個からなる環状ポリエン(シクロブタジエン)は極めて不安定である。電子求引性のペンタフルオロフェニル置換基を持つ置換シクロブタジエンは、最低空分子軌道のエネルギーが下がっているため、アセチレンやエチレンとのディールス・アルダー反応はもとより、ベンゼンとのディールス・アルダー反応においても、ジエンとしての反応性が著しく高まっている。今回我々は、こうした置換シクロブタジエンとベンゼンとの反応が120℃という比較的低温で起こり、ユニークなディールス・アルダー/レトロ・ディールス・アルダー反応を経てベンゼンがC2H2ユニットとC4H4ユニットに形式的に分解されることを示している。この結果は、遷移金属による活性化を行わなくてもベンゼンのC-C結合を切断できる珍しいケースの新たな一例である。
中本 真晃1, 稲垣 佑亮1 & 関口 章1
- 筑波大学 数理物質系 化学域
Cyclic polyene with six carbon atoms (benzene) is very stable, whereas cyclic polyene with four carbon atoms (cyclobutadiene) is extremely unstable. The electron-withdrawing pentafluorophenyl group of a substituted cyclobutadiene lowers the energy of the lowest unoccupied molecular orbital, greatly increasing its reactivity as a diene in Diels–Alder reactions with acetylene, ethylene and even benzene. Here we show that the reaction with benzene occurs cleanly at the relatively low temperature of 120 °C and results in the formal fragmentation of benzene into C2H2 and C4H4 units, via a unique Diels–Alder/retro-Diels–Alder reaction. This is a new example of the rare case where breaking the C–C bond of benzene is possible with no activation by a transition metal.

