有機ナトリウム化合物による触媒的クロスカップリング
Organosodium compounds for catalytic cross-coupling
2019年3月18日 Nature Catalysis 2 : 3 doi: 10.1038/s41929-019-0250-6
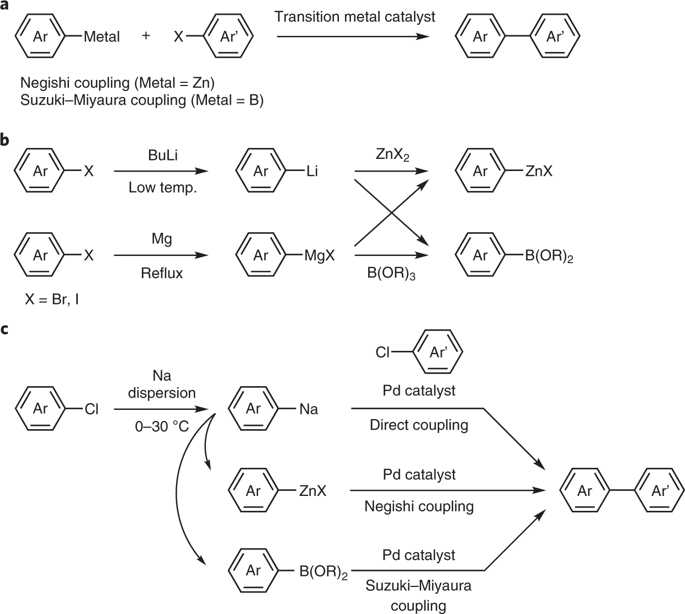
ナトリウムは、地球の地殻と海洋に最も豊富に存在するアルカリ金属である。しかし、有機ナトリウム化合物は、前世紀の合成有機化学で主流であった有機リチウム化合物に劣ると長い間考えられていた。有機ナトリウム化合物はその高い反応性のためにほとんど無視されてきたが、リチウムなどの存在量が少ない元素に頼らない持続可能な合成がますます求められていることを考慮すると、有機ナトリウム化学は再検討する価値がある。今回我々は、通説に反して、有機ナトリウム化合物が塩化アリールや(ヘテロ)アレーンと扱いやすいナトリウム分散体から容易に合成でき、対応する亜鉛化合物やホウ素化合物にトランスメタル化した後、有機合成における基本的な炭素–炭素結合形成反応である根岸クロスカップリング反応や鈴木・宮浦クロスカップリング反応に容易に利用できることを実証した。さらに、有機ナトリウム種との直接カップリング反応も可能であった。
Corresponding Author
Sodium is the most abundant alkali metal in the Earth’s crust and the ocean. However, organosodium compounds have long been considered inferior to organolithium compounds, which have instead dominated synthetic organic chemistry during the last century. Despite being largely neglected because of their reactive nature, it is worth re-exploring organosodium chemistry, in light of the growing demand for sustainable syntheses without recourse to less abundant elements such as lithium. Herein, we demonstrate that, contrary to common belief, organosodium compounds can be easily prepared from aryl chlorides or (hetero)arenes and easy-to-handle sodium dispersion and, after being transmetallated to the corresponding zinc and boron compounds, they readily participate in the Negishi and Suzuki–Miyaura cross-coupling reactions, fundamental carbon–carbon bond-forming reactions in organic synthesis. Direct coupling reactions with organosodium species were also possible.

