Understanding longevity: From gene sequences to social inequity
03 April 2025
Published online 13 May 2021
A chance discovery reveals a potential strategy for engineering cellular protein manufacturing as a means to boost biotech productivity.
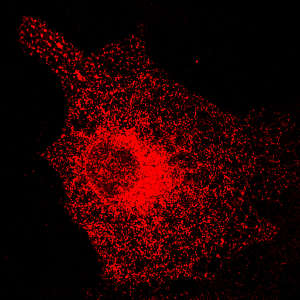
Dr. Despina Kerselidou, Maastricht University Nanoscopy Core facility, Netherlands
A researcher at New York University Abu Dhabi, Kourosh Salehi-Ashtiani, and his student, Sarah Daakour, initially set out to identify proteins involved in a particular form of leukaemia. After homing in on a glycosylation enzyme called exostosin-1 (EXT1), they teamed up with Franck Dequiedt and Jean-Claude Twizere at the University of Liege in Belgium to assess how this protein affects white blood cell development. As the researchers began manipulating EXT1 levels in cultured cells, they noticed surprising perturbations in an organelle called the endoplasmic reticulum (ER).
The ER is a set of stacked, membrane-bound structures that act as an assembly line for the processing and enzymatic modification—including glycosylation—of newly synthesized proteins. When Salehi-Ashtiani and colleagues experimentally knocked down expression of EXT1, they noticed a clear change in the structure of the ER, which became abnormally elongated relative to unmodified cells. Subsequent experiments showed that EXT1 regulates the function and stability of other key components of the ER glycosylation machinery, which in turn govern the structure and organization of this organelle.
The researchers also observed striking differences in the lipid composition and physical properties of the ER membrane, and a boost in the rate at which proteins processed in the ER were shuttled to the cell membrane.
Salehi-Ashtiani was taken aback by the far-reaching effects of this one glycosylation enzyme on ER function. “The functional consequences highlighted in our article… illustrate the remarkable research opportunities for exploiting the structural diversity of glycans and their processing enzymes, such as EXT1,” he says.
These findings are not just of academic interest. Cultured cells are used as factories for producing a wide range of protein-based drugs and vaccines, and ER processing is a core component of this biomanufacturing process. “We showed that by controlling the expression of EXT1, this production line is extended and is more productive than in control cells,” says Salehi-Ashtiani. “We envision that our findings can be used to develop cell lines for enhanced production of complex recombinant proteins and viral vectors, such as the highly-needed adenovirus-based SARS-CoV-2 vaccines.”
His team is now exploring this potential, while also continuing to explore the broader impact of glycosylation on the biological function of both healthy and cancerous cells.
doi:10.1038/nmiddleeast.2021.45
Kerselidou, D. et al. Alternative glycosylation controls endoplasmic reticulum dynamics and tubular extension in mammalian cells. Sci. Adv. 7, eabe8349 (2021).
Stay connected: