ニッケルによる不斉非対称化を経由するフェノール類からの縮合三環式骨格の2段階合成
Two-step synthesis of chiral fused tricyclic scaffolds from phenols via desymmetrization on nickel
2017年6月26日 Nature Communications 8 : 32 doi: 10.1038/s41467-017-00068-8
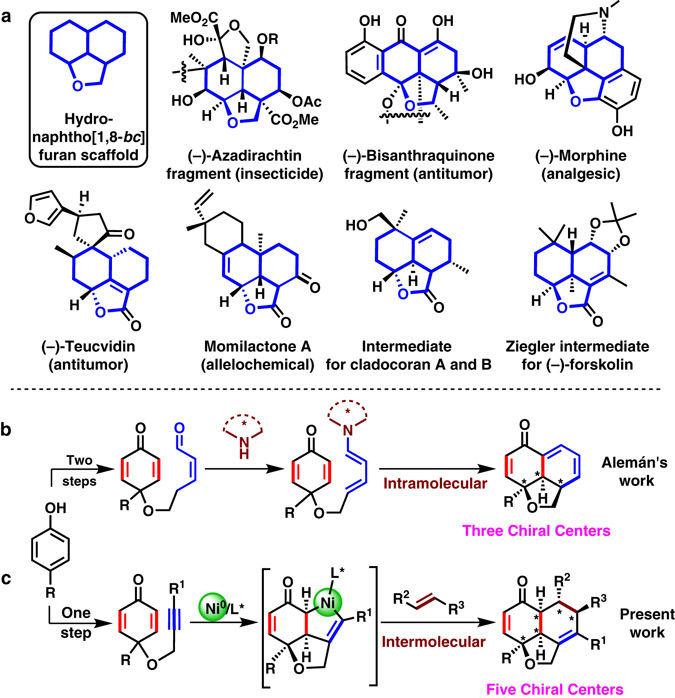
複数の不斉炭素中心を持つ三環式フラン誘導体は天然物中によく見られる骨格である。当該三環式骨格を立体選択的かつ廃棄物を出さずに迅速合成する手法の開発は重要な課題である。本研究では連続した5つの不斉炭素を含むヒドロナフト[1.8-bc]フランのエナンチオ選択的合成を達成した。本反応の鍵過程はニッケル上での酸化的環化によるアルキニルシクロヘキサジエノンの非対称化と続く[4+2]環状付加反応である。アルキニルシクロヘキサジエノンは容易に入手可能なフェノール類から1段階で合成した。本反応は高い化学選択性、位置選択性、そしてエナンチオ選択性を示す。また、[4+2]環状付加の過程におけるエノンの挿入に関する化学選択性は従来の報告と逆であり、本研究により環状ニッケル錯体の新規な反応性が明らかになった。鍵となる非対称化過程は、化学量論反応による環状ニッケル錯体の単離により確認した。生成物であるヒドロナフト[1.8-bc]フラン誘導体はC=OやC=Cなどの官能基を含んでおり、これらを誘導体化することによってより多様な三環式フラン誘導体を合成した。
Corresponding Author
Tricyclic furan derivatives with multiple chiral centers are ubiquitous in natural products. Construction of such tricyclic scaffolds in a stereocontrolled, step-economic, and atom-economic manner is a key challenge. Here we show a nickel-catalyzed highly enantioselective synthesis of hydronaphtho[1,8-bc]furans with five contiguous chiral centers via desymmetrization of alkynyl-cyclohexadienone by oxidative cyclization and following formal [4 + 2] cycloaddition processes. Alkynyl-cyclohexadienone was synthesized in one step from easily accessible phenols. This reaction represents excellent chemo-selectivity, regio-selectivity, diastereo-selectivity, and enantio-selectivity (single diastereomer, up to 99% ee). An extraordinary regioselectivity in the formal [4 + 2] cycloaddition step with enones revealed the diverse reactivity of the nickelacycle intermediate. Desymmetrization of alkynyl-cyclohexadienones via oxidative cyclization on nickel was supported by the isolation of a nickelacycle from a stoichiometric reaction. Enantioenriched tricyclic products contain various functional groups such as C=O and C=C. The synthetic utility of these products was demonstrated by derivatization of these functional groups.

