Research Abstract
Mg2+チャネルMgtEの高分解能結晶構造から明らかになったイオン選択性の構造基盤
Structural basis for ion selectivity revealed by high-resolution crystal structure of Mg2+ channel MgtE
2014年11月4日 Nature Communications 5 : 5374 doi: 10.1038/ncomms6374
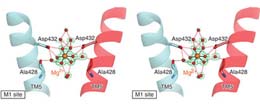
マグネシウムは生細胞中に最も豊富に存在する2価陽イオンで、複数の生物学的過程で非常に重要な役割を担っている。MgtEは真核生物,細菌,古細菌にわたり広く分布しているMg2+チャネルで、細胞内Mg2+の恒常性維持に関わっている。本論文では、MgtEの膜貫通ドメインについて、Mg2+、Mn2+、あるいはCa2+が結合した状態の高分解能結晶構造を報告する。Mg2+が結合している状態の高分解能結晶構造では、選択フィルター内にある水和したMg2+がはっきり見られる。これらの構造と生化学的解析に基づいて、我々は、完全に水和したMg2+イオンの水和殻の形が選択性フィルター中のカルボン酸側鎖によって認識されるという、MgtEの陽イオン選択機構を提案する。これは脱水和したK+イオンが認識される、KcsAのK+選択性フィルターとは対照的である。我々の結果からはまた、ペリプラズム側にある陽イオン結合部位が明らかになった。これはチャネルの開口を調節し,Mg2+に類似した陽イオンの通過を妨げている。
竹田 弘法1,2, 服部 素之1,2,3,4, 西澤 知宏1,2, 山下 恵太郎5, Syed T. A. Shah6, Martin Caffrey6, Andrés D. Maturana7, 石谷 隆一郎1,2 & 濡木 理1,2
- 東京大学大学院 理学系研究科 生物科学専攻
- 理化学研究所 グローバル研究クラスタ
- 科学技術振興機構 さきがけ
- 復旦大学(中国)
- 理化学研究所 放射光科学総合研究センター 生命系放射光利用システム開発ユニット
- ダブリン大学 トリニティカレッジ(アイルランド)
- 名古屋大学大学院 生命農学研究科 生命技術科学専攻
Magnesium is the most abundant divalent cation in living cells and is crucial to several biological processes. MgtE is a Mg2+ channel distributed in all domains of life that contributes to the maintenance of cellular Mg2+ homeostasis. Here we report the high-resolution crystal structures of the transmembrane domain of MgtE, bound to Mg2+, Mn2+ and Ca2+. The high-resolution Mg2+-bound crystal structure clearly visualized the hydrated Mg2+ ion within its selectivity filter. Based on those structures and biochemical analyses, we propose a cation selectivity mechanism for MgtE in which the geometry of the hydration shell of the fully hydrated Mg2+ ion is recognized by the side-chain carboxylate groups in the selectivity filter. This is in contrast to the K+-selective filter of KcsA, which recognizes a dehydrated K+ ion. Our results further revealed a cation-binding site on the periplasmic side, which regulate channel opening and prevents conduction of near-cognate cations.

