Research Abstract
無機エレクトライド表面における二酸化炭素の活性化および分解
Activation and splitting of carbon dioxide on the surface of an inorganic electride material
2013年8月29日 Nature Communications 4 : 2378 doi: 10.1038/ncomms3378
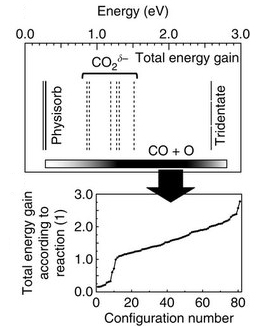
二酸化炭素の活性化は、二酸化炭素を有用化学物質に変換する過程において最も重要である。二酸化炭素の活性化には、仕事関数の低い安定な酸化物の表面が有望だと考えられる。今回我々は、無機エレクトライド[Ca24Al28O64]4+(e−)4の表面が室温で二酸化炭素を活性化し分解することを報告する。この挙動は、表面に近い領域で局在化した電子の濃度が高いことと、酸素原子とひずんだ一酸化炭素および二酸化炭素分子が表面の凹凸部に捕捉されることに起因する。ここでは、昇温脱離法と分光学的手法を用いて、二酸化炭素に暴露した[Ca24Al28O64]4+(e−)4表面を調査した。第一原理計算によって裏付けられた測定結果から、一酸化炭素と二酸化炭素の両方が室温以上で[Ca24Al28O64]4+(e−)4表面に特異な配置で吸着して、加熱すると一酸化炭素分子と原子状酸素が脱離することが明らかになっている。
戸田 喜丈1, 平山 博之2, Navaratnarajah Kuganathan3, Antonio Torrisi3, Peter V. Sushko3 & 細野 秀雄1, 4
- 東京工業大学 フロンティア研究機構
- 東京工業大学大学院 総合理工学研究科 材料物理科学専攻
- ロンドン大学ユニバーシティカレッジ(英国)
- 東京工業大学 元素戦略研究センター
Activation of carbon dioxide is the most important step in its conversion into valuable chemicals. Surfaces of stable oxide with a low work function may be promising for this purpose. Here we report that the surfaces of the inorganic electride [Ca24Al28O64]4+(e−)4 activate and split carbon dioxide at room temperature. This behaviour is attributed to a high concentration of localized electrons in the near-surface region and a corrugation of the surface that can trap oxygen atoms and strained carbon monoxide and carbon dioxide molecules. The [Ca24Al28O64]4+(e−)4 surface exposed to carbon dioxide is studied using temperature-programmed desorption, and spectroscopic methods. The results of these measurements, corroborated with ab initio simulations, show that both carbon monoxide and carbon dioxide adsorb on the [Ca24Al28O64]4+(e−)4 surface at RT and above and adopt unusual configurations that result in desorption of molecular carbon monoxide and atomic oxygen upon heating.

