Research Abstract
腸内細菌叢はインスリンの作用による脂肪の蓄積を短鎖脂肪酸受容体GPR43を介して抑制する
The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43
2013年5月7日 Nature Communications 4 : 1829 doi: 10.1038/ncomms2852
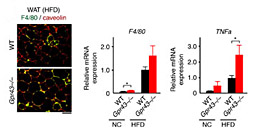
腸内細菌叢は、宿主の栄養獲得およびエネルギー調節に影響し、肥満、インスリン抵抗性、糖尿病の発生に影響を及ぼしている可能性がある。食事時、腸内細菌群は、宿主にとって重要なエネルギー源である短鎖脂肪酸を産生する。本論文では、短鎖脂肪酸受容体GPR43が、腸内細菌叢の代謝活性と宿主の体のエネルギー恒常性維持を結びつけることを報告する。GPR43欠損マウスは通常の餌で肥満となるが、脂肪組織でGPR43を特異的に過剰発現させたマウスは高脂肪食を与えた場合でも痩せたままであった。無菌条件で生育させるか、あるいは抗生物質処置後には、いずれのマウスとも表現型は正常であった。また、短鎖脂肪酸によるGPR43の活性化が、脂肪細胞ではインスリンシグナル伝達を抑制することで脂肪組織における脂肪蓄積を抑制し、他の組織では、取り込まれていない脂質およびグルコースの代謝を促進することも明らかとなった。これらの結果から、GPR43が過剰な食事性エネルギーに対するセンサーとして機能し、代謝の恒常性を維持しながら体内エネルギー利用を制御していることが分かる。
木村 郁夫1, 小澤 健太郎2, 井上 大輔1, 今村 武史3, 木村 久美4, 前田 岳志1, 寺澤 和哉1, 柏原 大二1, 平野 加奈子1, 谷 妙子1, 高橋 知行1, 宮内 諭1, 塩井 剛5, 井上 啓4 & 辻本 豪三1,6
- 京都大学大学院 薬学研究科 薬理ゲノミクス
- 奈良県立医科大学 薬理学部
- 滋賀医科大学 薬理学部
- 金沢大学 脳・肝インターフェイスメディシン研究センター 生体統御学部門(代謝生理学)
- 理化学研究所 発生・再生科学総合研究センター 動物資源開発室
- 京都大学大学院 薬学研究科 ゲノム創薬科学
The gut microbiota affects nutrient acquisition and energy regulation of the host, and can influence the development of obesity, insulin resistance, and diabetes. During feeding, gut microbes produce short-chain fatty acids, which are important energy sources for the host. Here we show that the short-chain fatty acid receptor GPR43 links the metabolic activity of the gut microbiota with host body energy homoeostasis. We demonstrate that GPR43-deficient mice are obese on a normal diet, whereas mice overexpressing GPR43 specifically in adipose tissue remain lean even when fed a high-fat diet. Raised under germ-free conditions or after treatment with antibiotics, both types of mice have a normal phenotype. We further show that short-chain fatty acid-mediated activation of GPR43 suppresses insulin signalling in adipocytes, which inhibits fat accumulation in adipose tissue and promotes the metabolism of unincorporated lipids and glucose in other tissues. These findings establish GPR43 as a sensor for excessive dietary energy, thereby controlling body energy utilization while maintaining metabolic homoeostasis.

