Research Abstract
アキラル成分を用いた2回らせん水素結合ネットワークにおける分子キラリティーと超分子キラリティーの間のつながり制御
Linkage control between molecular and supramolecular chirality in 21-helical hydrogen-bonded networks using achiral components
2013年4月30日 Nature Communications 4 : 1787 doi: 10.1038/ncomms2756
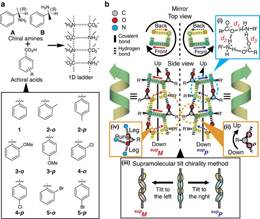
キラル分子は、分子の絶対配置を反映して片方のキラリティーの超分子集合体を優先的に形成する。しかし、特定の条件下では、結合長の柔軟性と非共有結合相互作用の可逆性のために、キラリティーが逆の超分子集合体も得られる。このキラリティーの選択性やスイッチング現象の機構はまだよくわかっておらず、ほとんどの現象は偶然観察される。今回我々は、結晶学的研究に基づいて結晶状態でのキラリティーを制御した、キラルな水素結合2回らせん集合体の構造構築を実証する。得られた結晶構造を詳細に調べることで、キラリティーのスイッチング機構を明らかにし、これに基づきアキラルな要素を導入して超分子集合体のキラリティーの制御に成功した。この研究によって、結晶状態における分子キラリティーと超分子キラリティーのつながりが明確になった。
佐々木 俊之1, 久木 一朗1, 宮野 哲也1, 藤内 謙光1, 森本 和也2, 佐藤 久子2, 都築 誠二3 & 宮田 幹二1
- 大阪大学大学院 工学研究科 生命先端工学専攻
- 愛媛大学大学院 理工学研究科 環境機能科学専攻
- 独立行政法人 産業技術総合研究所 ナノシステム研究部門 計算化学領域
Chiral molecules preferentially form one-handed supramolecular assemblies that reflect the absolute configuration of the molecules. Under specific conditions, however, the opposite-handed supramolecular assemblies are also obtained because of flexibility in the bond length and reversibility of non-covalent interactions. The mechanism of the handedness selectivity or switching phenomenon remains ambiguous, and most phenomena are observed by chance. Here we demonstrate the construction of chiral hydrogen-bonded twofold helical assemblies with controlled handedness in the crystalline state based on crystallographic studies. Detailed investigation of the obtained crystal structures enabled us to clarify the mechanism, and the handedness of the supramolecular chirality was successfully controlled by exploiting achiral factors. This study clearly reveals a connection between molecular chirality and supramolecular chirality in the crystalline state.

