Understanding longevity: From gene sequences to social inequity
03 April 2025
Published online 13 January 2020
The search for gene functions through multiple methods leads to the identification of a rare collagen-related syndrome.
Ela Knapik
Essential in connective tissue development and repair, collagen is the most abundant extracellular matrix protein in vertebrates. Genetic defects affecting collagen’s intracellular trafficking and secretion can lead to impaired extracellular matrix formation and related diseases, but the details of these processes have been unclear.
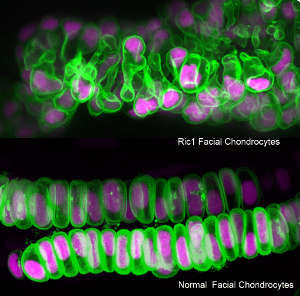
Researchers focused on Ric1, a protein that is highly evolutionarily conserved between humans and zebrafish. They found that Ric1 interacts with other proteins to export the precursor of collagen outside the cell. Zebrafish lacking the ric1 gene have a small head, shortened trunk, and abnormally shaped cartilage cells.
In the search for the full spectrum of Ric1 functions, the researchers consulted the Vanderbilt University Medical Center’s BioVU Biobank, the UK Biobank and other publicly available datasets, to find the traits associated with a reduced amount of Ric1 protein in humans. This revealed the organs and systems that can be affected, helped to pinpoint core clinical features, and guided further zebrafish studies.
Supported by these data, Fowzan S. Alkuraya of the King Faisal Specialist Hospital and Research Center, Saudi Arabia, and colleagues re-examined eight young patients whose DNA has a double copy of a rare ric1 variant with a single mutation. These children took part in a previous study, led by the same researcher, about paediatric cataract. Beyond vision problems, the researchers took into consideration all their clinical features, naming a new syndrome: CATIFA, which involves cleft lip, cataract, tooth abnormality, intellectual disability, facial dysmorphism, and attention-deficit hyperactivity disorder. A skin fibroblast biopsy confirmed that Ric1’s molecular and cellular functions are identical in fish and man.
“Combining three sources of data has numerous advantages. Each helped refine the findings of the other two, and independently validated the results,” says Ela W. Knapik of Vanderbilt University Medical Center, USA, and leading author of the study.
The researchers applied the same triple approach successfully to two other genetic diseases of the connective tissue: cranio-lenticulo-sutural dysplasia and Marfan syndrome, for which clinical and animal model data are available.
“This approach holds the potential to hasten medical advancement in many poorly described hereditary skeletal diseases and disorders,” says geneticist Daniel Liedtke of the University of Würzburg, Germany, who was not involved in the study.
doi:10.1038/nmiddleeast.2020.4
Unlu, G. et al. Phenome-based approach identifies RIC1-linked Mendelian syndrome through zebrafish models, biobank associations and clinical studies. Nat. Med. https://doi.org/10.1038/s41591-019-0705-y (2020).
Stay connected: