The Missing Kingdom: Why Fungi Must Be Central to Conservation Strategy
28 December 2025
Published online 31 October 2018
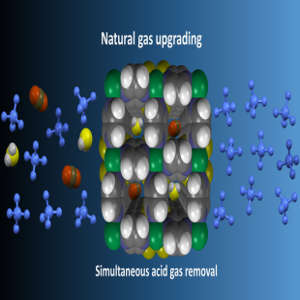
A porous solid-state material that can filter unwanted contaminants from natural gas could transform the production of gas-based fuels.
Mohamed Eddaoudi
Enlarge image
Now, Mohamed Eddaoudi, Youssef Belmabkhout and their team at King Abdullah University of Science and Technology (KAUST) in Saudi Arabia, together with scientists in France and the US, have designed and fabricated a fluorinated metal–organic framework (MOF) capable of filtering out both acid gases from natural gas, regardless of its composition and impurity concentrations.
MOFs are synthetic, solid-state materials constructed from networks of metal ions connected by organic linkers. Their structures are manipulated to fulfil specific tasks. When natural gas passes through the MOF constructed by Eddaoudi’s team, the CO2 and H2S molecules are attracted to and trapped by different nanoscale structures within its framework. The MOF performed effectively, removing the contaminants from natural gas streams at different pressures and temperatures.
Other MOF designs by the same team can single out contaminants for removal, offering multiple further applications.
“Recent advances in MOF chemistry by our KAUST team have permitted the design and construction of various MOF platforms appropriate for energy-intensive separations, such as natural gas upgrading,” says Eddaoudi.
doi:10.1038/nmiddleeast.2018.135
Belmabkhout, Y. et al. Natural gas upgrading using a fluorinated MOF with tuned H2S and CO2 absorption selectivity. Nat. Energy http://dx.doi.org/10.1038/s41560-018-0267-0 (2018).
Stay connected: