Understanding longevity: From gene sequences to social inequity
03 April 2025
Published online 12 September 2018
More efficient solar cells could be just one benefit of learning to control perovskite nanostructures.
Nature Materials 2018
“The promising light-harvesting and emissive properties of hybrid perovskites have captivated the optoelectronics research community,” says Rafael Quintero-Bermudez of the University of Toronto, whose team collaborated with researchers at Stanford University in the US and King Abdullah University of Science and Technology in Saudi Arabia. “Our study delves into the formation of these materials,” he says.
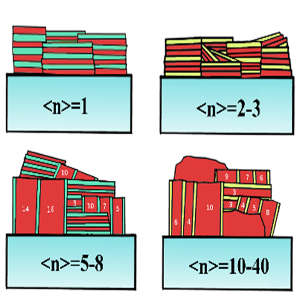
The work investigated reduced-dimensional metal halide perovskites (RDPs), which have two-dimensional layers containing metal ions and ions of halogen elements, especially chlorine, bromine and iodine. Adding organic (carbon-based) layers can lead to nanostructures that enhance the stability of the overall material.
The research uncovered how intermediate crystal fragments forming within a solvent mixture provide a scaffold that facilitates the crystallization of the desired RDPs. Varying the ions, solvents and deposition techniques allowed control of the layering of the RPD structure, thus changing its properties. X-ray scattering analysis provided further detail of the perovskite structure.
The findings should allow more precise control of RDP formation to create more efficient and longer-lasting solar cells, explains Quintero-Bermudez. They may also be more generally useful for making perovskites for other applications, including light-emitting diodes, transistors, catalysts, sensors and highly selective gas storage materials.
doi:10.1038/nmiddleeast.2018.107
Quintero-Bermudez, R., et al. Compositional and orientational control in metal halide perovskites of reduced dimensionality. Nat. Mater. https://doi.org/10.1038/s41563-018-0154-x (2018).
Stay connected: