Understanding longevity: From gene sequences to social inequity
03 April 2025
Published online 22 May 2017
Two teams of Arab and American researchers are ‘tantalizingly close’ to generating primordial blood stem cells in the lab.

© Rio Sugimura
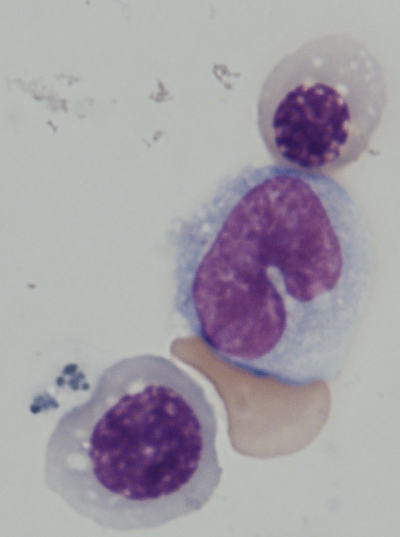
Scientists have previously succeeded in genetically reprogramming skin cells to make pluripotent stem (iPS) cells, which are later used to generate multiple human cell types. However, the ability to induce blood stem cells that self-regenerate, for the treatment of millions affected by blood cancers and genetic disorders, has eluded researchers.
The two papers newly published in Nature describe methods that pave the way for safe, artificial and bona fide hematopoietic stem cells (HSCs) generation. Hematopoietic stem (HSC) cells are the common ancestor of all cells created in the body, producing billions of blood cells every day.
This bears major implications for cell therapy, drug screening and leukemia research. The root causes of blood diseases can be scrutinized and creating immune-matched blood cells, derived from a patient’s own cells, is now conceivable.
The first team, based at the Boston Children’s Hospital, has generated blood-forming stem cells (HSCs) in the lab using pluripotent stem cells for the first time.
“We’re tantalizingly close to generating bona fide human blood stem cells in a dish,” says senior investigator George Daley, who heads the research lab in Boston Children’s Hospital’s stem cell program and who is dean of Harvard Medical School. “This work is the culmination of over 20 years of striving.”
Ryohichi ‘Rio’ Sugimura, the study’s first author and a postdoctoral fellow in the Daley Lab, says his team exposed human pluripotent stem cells (both ES and iPS cells) to chemical signals to prompt them to differentiate into specialized cells and tissues during embryonic development.
"Sugimura and his colleagues delivered transcription factors — proteins that control and regulate the transcription of specific genes — into the cells using a lentivirus, a vector to deliver genes. “The resultant cells were transplanted to immune deficient mice, where human blood and immune cells were made,” he says.
A few weeks after the transplant, a small number of rodents were found to be carrying multiple types of blood cells in their bone marrow and blood; cells that are also found in human blood. “This is a major step forward for our ability to investigate genetic blood disease,” says Daley.
The second team, a group of scientists from Weill Cornell Qatar and Weill Cornell Medicine in New York, used mature mouse endothelial cells — cells that line blood vessels — as their starting material for generating HSCs.
© Rio Sugimura
The team isolated the cells, and then pushed key transcription factors into their genomes. Between days 8 and 20 into the process, the cells specified and multiplied.
“Our research showed that endothelial cells can be converted into competent HSCs with the ability to both regenerate the myeloid and lymphoid lineage,” he explains.
The method brings hope for people afflicted with leukemia requiring HSCs transplantation, or genetic disorders affecting the myeloid or lymphoid lineages. The clinical generation of HSCs, derived from the same individual, can eventually help scientists correct genetic abnormalities.
As exciting as the two studies are, rigorous tests are still required to check the normality of lab-grown cells before the clinical phase, says Alexander Medvinsky, professor of hematopoietic stem cell biology at the University of Edinburgh Medical Research Council Centre for Regenerative Medicine. Medvinsky was not involved in either study.
“The risks of infusion of genetically engineered cells in humans should not be underestimated,” he weighs in. “Tests and trials to generate safe fully functional human blood stem cells may take many years, in contrast to similar assessment in short-living mice. It is not clear now whether blood stem cells can become cancerous in the longer term.”
He adds however that this “type of research is exactly what is required to potentially meet clinical needs.”
doi:10.1038/nmiddleeast.2017.89
Stay connected: