Understanding longevity: From gene sequences to social inequity
03 April 2025
Published online 16 September 2013
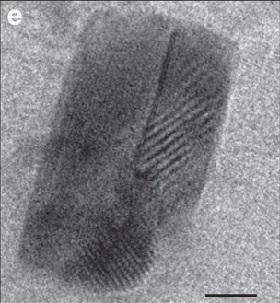
In low temperature aqueous solutions, mineral formation has been shown to be preceded by the formation of crystalline nanometre-sized particles. Yet this has not been shown to happen in hot fluid, or magmatic, systems. Now, a team of researchers, led by Hassan M. Helmy from Minia University in Egypt, has demonstrated for the first time that nanoparticles of noble metals can form in magmatic systems long before the solution precipitates crystals, publishing their research in Nature Communications1.
The researchers added traces of platinum and arsenic to a magmatic sulphide melt at a high temperature between 950°C and 1,180°C. Using high-resolution transmission electron microscopy, they found a wide range of Pt-As nanoparticles, ranging from poorly ordered clusters and platelets to nanometre-sized droplets of a Pt-enriched arsenide melt. The researchers suggest it would be easier for crystals of the trace mineral to form and grow on these nanoparticles, rather than forming from randomly spread anions and cations in the solution.
Helmy suggests their research could have far-reaching consequences on geochemistry. "Our experimental study demonstrates that highly siderophile (iron-loving) elements (HSE) form nano-associations in magmatic melts." In this form, their behaviour is governed by the surface properties and surface charges of these nanoparticles rather than the chemical properties of the elements as previously thought.
"Theories using HSE to quantify global and local differentiation processes in the Earth and HSE-ore forming processes should be revised according to the new fact," adds Helmy.
doi:10.1038/nmiddleeast.2013.156
Stay connected: