River subbasins more depleted than
official figures show
09 April 2024
Published online 27 July 2018
A healthy alpha-N-catenin protein is required for embryonic nerve fibres to reach their final destinations.
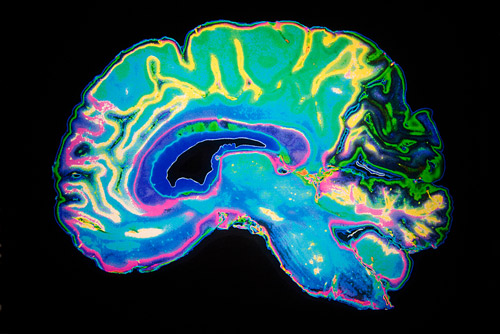
An international team of scientists, including from institutions in Egypt, Saudi Arabia, and Qatar, identified a mutation in three consanguineous families that causes a unique form of pachygyria. In this condition, the outer layer of the brain fails to fold in the way that gives it its usual cauliflower-shaped appearance. People with pachygyria have poor motor and cognitive development, undergo epileptic seizures, and severe intellectual impairment.
Joseph Gleeson of Howard Hughes Medical Institute at the University of California, San Diego and colleagues found affected members of the family had a mutation in both copies, each coming from one parent, of the CTNNA2 gene.
The mutation leads to malformation of a protein called alpha-N-catenin. Further investigations showed that the malformation affected how nerve fibres travel from their starting point in the developing brain to their final destination, a process called neuronal migration.
The team found that, in healthy brains, alpha-N-catenin binds to a filament-forming protein in nerve fibres, called actin, which is responsible for guiding nerve fibre microtubules by responding to extracellular cues. This binding prevents another protein complex, called ARP2/3, from binding to the actin. When alpha-N-catenin is malformed due to the CTNNA2 mutation, excessive amounts of ARP2/3 bind to actin, ultimately impairing the growth and stability of nerve cell projections.
“Our findings that alpha-N-catenin mutations cause pachygyria, probably by depressing Arp2/3-mediated actin branching, are an important first step toward understanding how neurons regulate their cytoskeleton during neuronal development,” says Ashleigh Schaffer, the first author of the study. The team is interested in further exploring this link in people with single-copy mutations of the gene who have epilepsy, autism and schizophrenia.
Geneticist, Fowzan Alkuraya, of King Faisal Specialist Hospital and Research Center in Saudi Arabia, who was not involved in the study says: “This brings immediate translational benefit in terms of prevention, not only to the study families, but any patient around the world carrying mutations in both copies of this newly identified disease gene.”
doi:10.1038/nmiddleeast.2018.85
Stay connected: