River subbasins more depleted than
official figures show
09 April 2024
Published online 26 July 2012
The formation of new blood vessels, angiogenesis, is regulated by the growth factor Vascular Endothelial Growth Factor-A (VEGF-A), which signals to endothelial cells by binding to various receptors (VEGFRs). This pathway must be tightly controlled; a slow down in VEGF-A activity can lead to pre-eclampsia, a dangerous form of hypertension that affects pregnant women. Overactive VEGF-A in muscles, on the other hand, can cause various vascular cancers.
A multinational team of scientists, led by Asif Ahmed of the University of Edinburgh, UK, and including Bahjat Al-Ani of King Khalid University, Abha, Saudi Arabia, investigated the diverse responses to VEGF-A signalling, publishing their findings in Nature Communications.
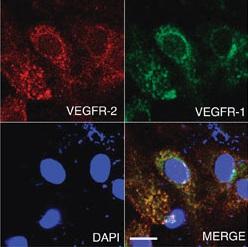
Receptor tyrosine kinases, such as the VEGFRs, are composed of pairs of subunits. In the cases of VEGFR-1 and VEGFR-2, the subunit of one receptor may pair with one subunit from the other, forming the heterodimer VEGFR1-2. However, it wasn't clear what function VEGFR1-2 has. By generating a dimeric ligand, an engineered molecule that can only activate VEGFR1-2, the researchers have been able to dissect the various roles of VEGF receptors in vitro.
VEGFR1-2 activation was found to control cell migration, tubular blood vessel formation and arterial relaxation, all VEGF-A activities. However, VEGFR1-2 did not induce cell proliferation or the production of endothelial tissue factors — roles instead mediated by homodimeric VEGFR-2. Activated VEGFR1-2 also inhibits various VEGFR-2 mediated functions.
The study suggests VEGFR-1 molecules, in general, will form VEGFR1-2 heterodimers, and roles previously ascribed to VEGFR-1 homodimers are actually the work of VEGFR1-2. Importantly, the study found that the signals sent by VEGFR1-2 and VEGFR-2 regulates and balances endothelial cell homeostasis and angiogenesis.
"There are new opportunities to investigate further the role of VEGF signalling in endothelial cell function," says Ahmed, who is hopeful that the engineered ligand could be one day be used as a therapeutic agent.
doi:10.1038/nmiddleeast.2012.105
Stay connected: